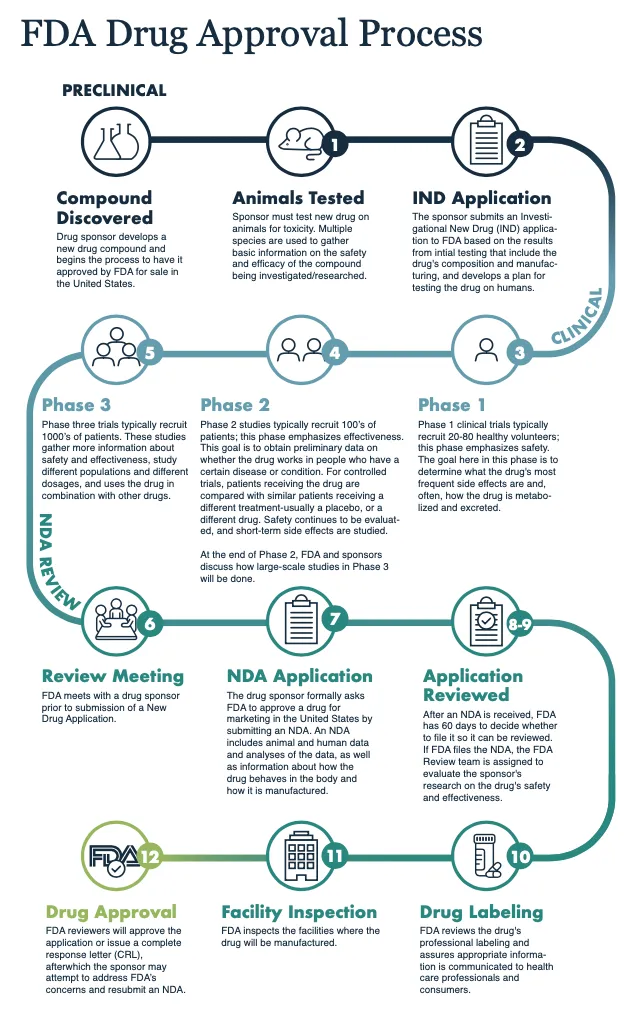
So now that you have a deeper sense of the complexity and time required to develop a new drug, what do you think the odds are that a new drug makes it all the way to your pharmacy’s shelf?
If you said “not high!” you’re right! Fewer than 10% of drugs that enter preclinical testing even make it into clinical trials. From there, approximately 63% of drugs that enter Phase I trials move on to Phase II, and about 30% of those drugs progress to Phase III. Once in Phase III, the success rate is higher; 58% of drugs at this stage win FDA approval. (Source: Clinical Development Success Rates 2006-2015 )
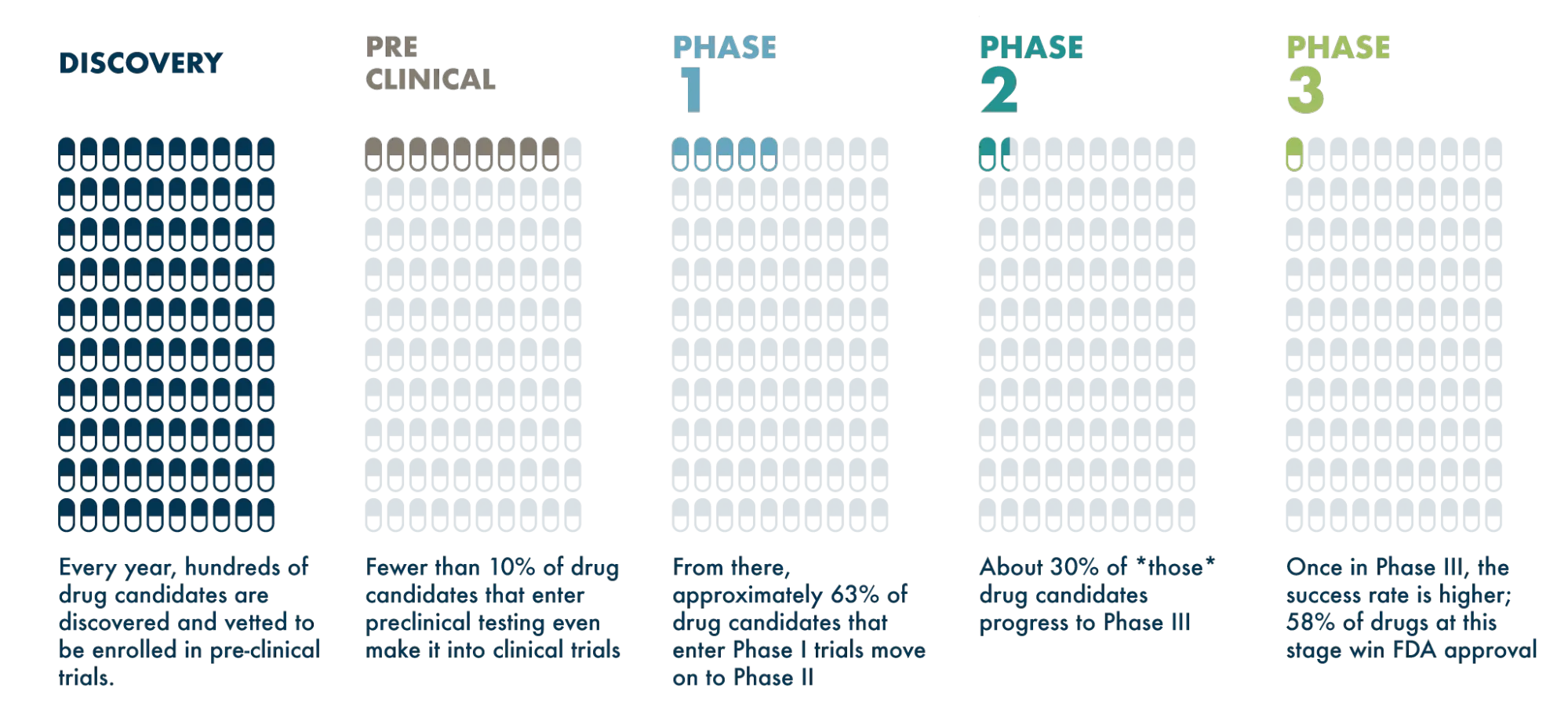
So when we think about the entire process, the overall success rate for a drug from discovery to FDA approval is only around 1%!