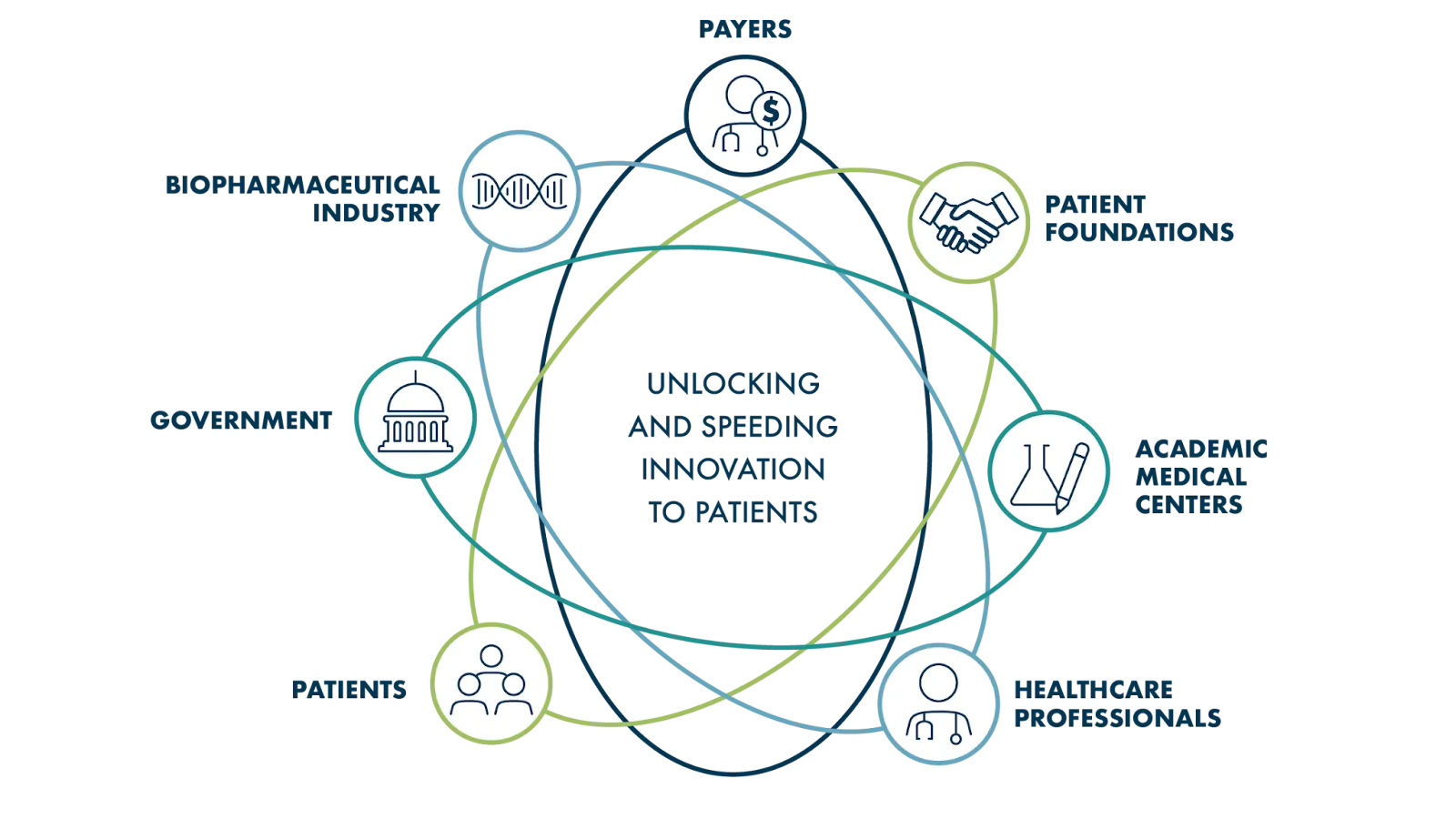
So if drug development is so expensive and most drug candidates fail before making it to patients, where does the cash to run this process come from? Our next chapter covers what you’re probably curious about: the money! But first, if you’d like to go a bit deeper into drug development, we recommend this truly fascinating [OPTIONAL] article: Operation Warp Speed: The Untold Story of the COVID-19 Vaccine, which appeared in Vanity Fair in November 2023. The first vaccines for COVID-19 were developed at record-breaking speed, and the article shares the incredible and intense story behind how that occurred. It also illuminates the complex interplay between politics, science, finance, manufacturing, and regulation that will always be at work in drug development.

Yes, we’ve just finished telling you about how long new drug development usually takes. But the COVID-19 vaccine approval process represents an exception to these norms—it essentially demonstrates the maximum fastest possible timeline. Several factors contributed to this success: assembly of an incredible team of the very best people; massive public and government support; significant prevalence of the disease among all ages and ethnicities all over the world; emergency authorization of the first products by the FDA; and the fact that money was no object on our way to a solution.